BRIEF DESCRIPTION
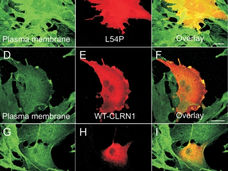
CLRN1 is a type of retinitis pigmentosa (RP) caused by mutations in the CLRN1 gene. The CLRN1 gene is thought to play a role in communication between nerve cells in the retina and inner ear. It may also be involved in the development and function of synapses, the junctions between neurons. The CLRN1 gene produces the clarin 1 protein, which is associated with hair cells in the inner ear and photoreceptors in the retina. Mutations in CLRN1 can alter the clarin 1 protein, causing it to be abnormally short or nonfunctional. This can lead to a loss of hair cells in the inner ear, which are responsible for transmitting sound and signals to the brain. Mutations in the CLRN1 gene are a primary cause of Usher syndrome type III (USH3), a condition that leads to progressive hearing loss and vision loss. In addition to hearing and vision loss, people with Usher syndrome may also have balance and coordination problems. The prevalence of Usher syndrome varies by country, but it's a significant cause of hearing loss in children. In the United States, it affects up to 16.7 per 100,000 children.
At present, there's no cure for this form of retinitis pigmentosa. For some forms of RP, medications can help treat complications. A medical professional can diagnose and manage the condition to address symptoms.
CLRN1
Disease Category: autosomal recessive
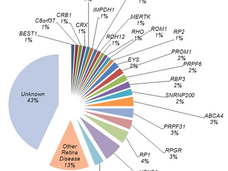
Patient Population:
Known Clinical Trials: None known
Treatment Options: Cochlear Implants (hearing)
Strategies to Preserve Eye Health: lutein
Institution(s) Conducting Research: Univ of Edinburgh, Royal Free Hospital (London), Univ of Michigan, MRC Institute of Genetics and Molecular Medicine (Edinburgh), Princess Alexandra Eye Pavilion (Edinburgh)
A FACE OF RP

tbd
tbd